|
mostly all textbooks on the spectral lines of elements tend to only go up to uranium, however, textbooks go into much more detail for each element. You can see a list of of all the known spectral lines of elements on Wikipedia since it seems to have the most updated table. These heavier elements likely have their own spectral lines, however, due to all I stated above, it isn't exactly possible to measure. This isn't even considering how much this would cost. Atoms of individual elements emit light at only specific wavelengths, producing a line spectrum rather than the continuous spectrum of all wavelengths produced by a hot object. This would make measuring their spectral lines nearly impossible. This produces an absorption spectrum, which has dark lines in the same position as the bright lines in the emission spectrum of an element. 
It's incredible because some of the heavier and more unstable elements have insanely short half lives ranging from 100.5 days (the most stable isotope of fermium (Fm, 100)), to 0.69 microseconds (0.00069 milliseconds) (Oganesson (Og, 118)). The electrons that drop from the higher energy levels to the lower energy levels in an atom release a photon with a specific wavelength, which generates the atomic emission spectrum. I could not find any data as to why this may be, however, I believe it is simply because we could not test for the spectral lines of the heavier elements due to their instability and scarcity. Explain how the atomic emission spectra occur and how they relate to the elements on the periodic table. Also, tables of the spectral lines of elements only seem to go up to the 99th element, Einsteinium (not including Astatine (At, 85) and Francium (Fr, 87). To measure these wavelengths in the laboratory, we must first separate them. Thus, the spectrum of an element can be stated by listing the particular wavelengths of light that its atoms emit. However, it is much more difficult to observe the spectral lines of liquids and solids due to how close together the atoms are. These two relationships combine to give a third: E hc (14A.3) (14A.3) E h c. Each thin band in each spectrum corresponds to a single, unique transition between energy levels in an atom. 5: The atomic emission spectra for various elements. 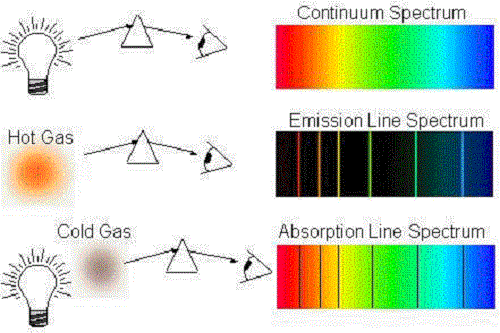
Each element has its own distinct spectral line and this fact can and has been used to identify an element. Scientists needed a fundamental change in their way of thinking about the electronic structure of atoms to advance beyond the Bohr model. Actually, this phenomena can be observed with liquids and solids as well. As the energy levels of the atom are quantized, the spectrum formed will contain wavelengths that will further reflect the differences in the energy levels.It's a good question, it's not stupid. On the other hand, when a hydrogen atom produce or emit a photon, the electron thus undergoes a transition from a higher energy level to a lower energy state (for instance n = 4 n = 3) During this transition that is from a higher energy level to a lower energy level, a transmission of light occurs. When a hydrogen atom absorbs a photon then the energy of the photon causes the electron to undergo a transition to a higher energy level (for instance n = 2 n = 3). The electromagnetic spectrum is a continuous distribution of wavelengths ranging from ultraviolet to infrared radiation.The hydrogen emission spectrum generally comprises of radiations of distinct frequencies. When an electric discharge is passed through a gaseous hydrogen molecule, it was found that the hydrogen atoms present in the molecule dissociate thus leading to the emission of electromagnetic radiation by the excited hydrogen atoms. If the light is passed through a prism or diffraction grating, it is split into its various colors. 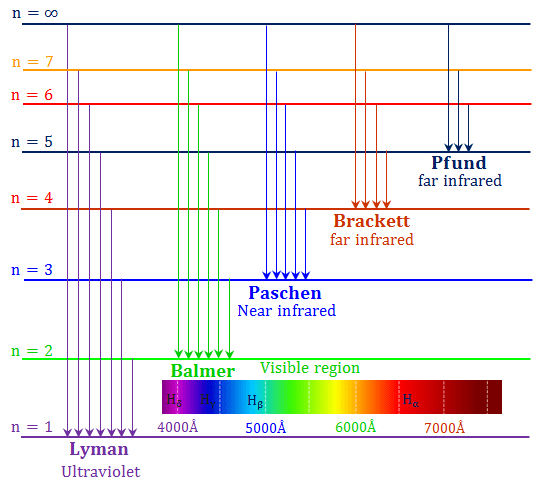
If a high voltage (5000 volts) is applied, the tube lights up with a bright pink glow.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
